|
As this chemical knowledge expanded rapidly, the need for classification became evident. List of ElementsĮvolution of Periodic Law and Classificationĭuring the early 19th century, analytical chemistry experienced significant advancements, leading to a vast accumulation of knowledge about the properties of elements and compounds. Various alternative representations of the periodic law exist, and discussions persist regarding the optimal form of the periodic table. Additionally, ongoing scientific discourse explores the correct positioning of certain elements within the present-day table. The extent to which the table will expand beyond these seven rows and whether the patterns observed in the known region will persist in this uncharted territory remain unknown. However, further research is required to fully characterize the chemical properties of the heaviest elements and validate their assigned positions. Currently, all 118 known elements, completing the first seven rows of the table, have been identified. Naturally occurring elements exist up to atomic number 94, necessitating the synthesis of new elements in laboratory settings to extend beyond this limit. Since then, the periodic table and the periodic law have become essential and indispensable components of modern chemistry.Īs scientific knowledge progresses, the periodic table continues to evolve. Seaborg in 1945, clarifying that the actinides belonged to the f-block rather than the d-block elements. The modern form of the table, as we recognize it today, was achieved with the discovery made by Glenn T. 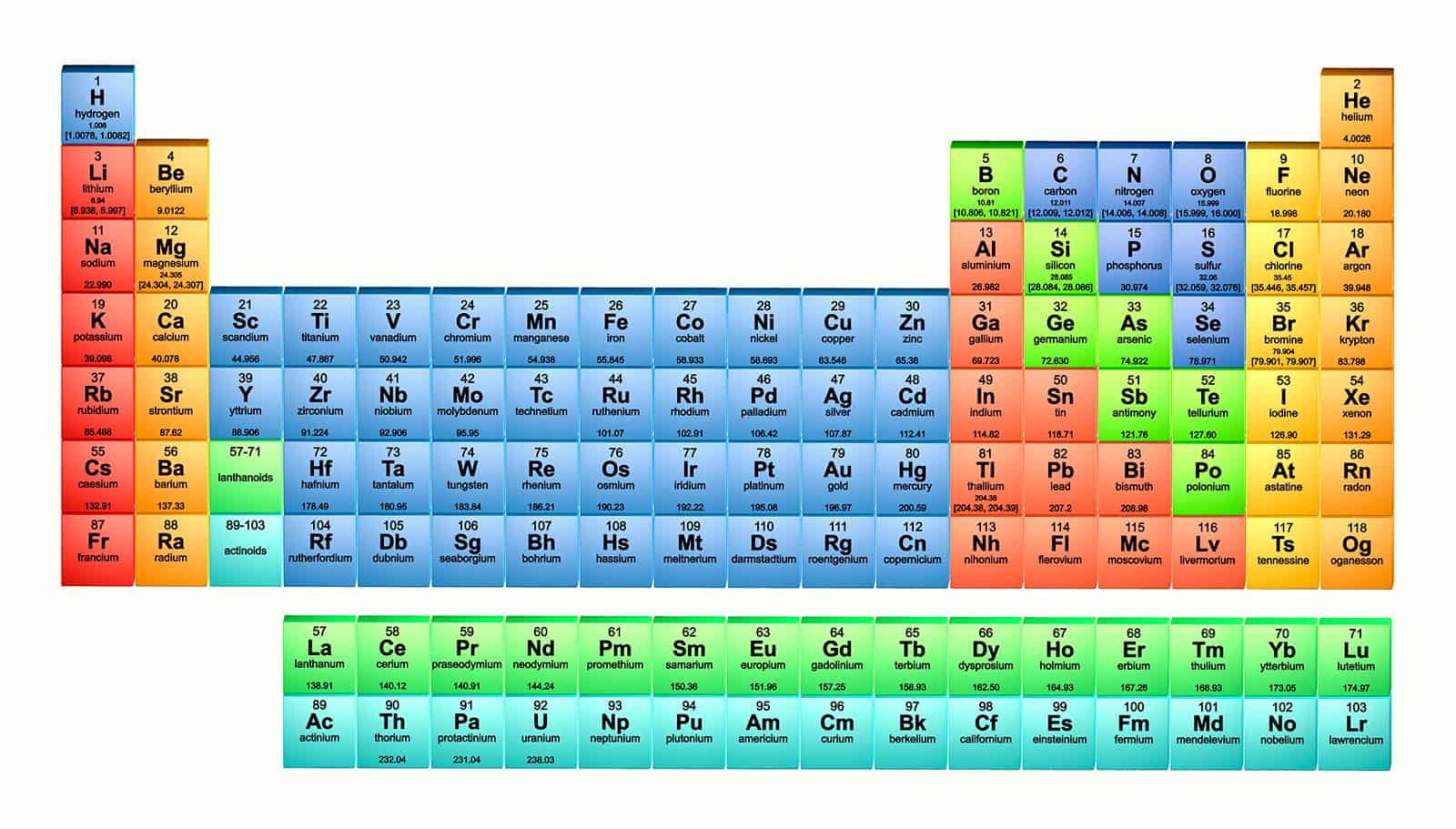
The periodic law became a significant discovery in the late 19th century and was later explained through the identification of atomic numbers and advancements in quantum mechanics during the early 20th century, which illuminated the internal structure of atoms. However, Mendeleev successfully utilized the periodic law to predict the properties of some of these missing elements. At the time, not all elements were known, resulting in gaps within the table. Mendeleev’s periodic table established the periodic law by highlighting the dependence of chemical properties on atomic mass. The earliest widely accepted version of the periodic table was formulated by the Russian chemist Dmitri Mendeleev in 1869. Other atomic variations, such as nuclides and isotopes, are categorized in separate tables like the tables of nuclides, often known as Segrè charts. 
The periodic table exclusively features electrically neutral atoms, where the number of positively charged protons matches the number of negatively charged electrons, and it also accommodates isotopes, which have the same number of protons but varying numbers of neutrons. These trends are attributed to the electron configurations of atoms. Notable trends can be observed across the table, such as the increase in nonmetallic character from left to right along a period, and from bottom to top within a group, while metallic character follows the opposite trend. Divided into four main blocks, the table consists of rows known as periods and columns known as groups.Įlements within the same group share similar chemical characteristics. The periodic table visually represents the periodic law, which states that the properties of elements exhibit a recurring pattern based on their atomic numbers. It serves as a fundamental tool in various scientific disciplines, particularly chemistry and physics, and is widely recognized as an emblem of the field of chemistry. The periodic table, also referred to as the periodic table of elements, is a systematic arrangement of chemical elements organized in rows and columns. Basic Details of Periodic Table of Elements
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
